English: First few
hydrogen atom orbitals; cross section showing color-coded probability density for different n=1,2,3 and l="s","p","d"; note: m=0
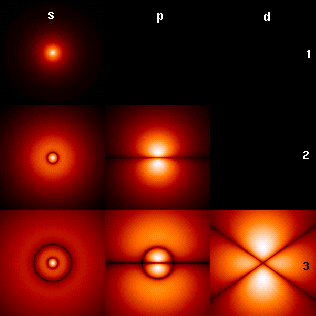
The picture shows the first few hydrogen atom orbitals (energy eigenfunctions). These are cross-sections of the probability density that are color-coded (black=zero density, white=highest density). The angular momentum quantum number l is denoted in each column, using the usual spectroscopic letter code ("s" means l=0; "p": l=1; "d": l=2). The main quantum number n (=1,2,3,...) is marked to the right of each row. For all pictures the magnetic quantum number m has been set to 0, and the cross-sectional plane is the x-z plane (z is the vertical axis). The probability density in three-dimensional space is obtained by rotating the one shown here around the z-axis.
Note the striking similarity of this picture to the diagrams of the normal modes of displacement of a soap film membrane oscillating on a disk bound by a wire frame. See, e.g.,
Vibrations and Waves, A.P. French, M.I.T. Introductory Physics Series, 1971,
ISBN 0393099369, page 186, Fig. 6-13. See also
Normal vibration modes of a circular membrane.
Slovenščina: Valovne funkcije elektrona v
vodikovem atomu imajo določeno
energijo (naraščajoče od zgoraj:
n=1,2,3 ...) in
vrtilno količino (naraščajoče prek:
s,
p,
d ...). Svetlejša področja odgovarjajo višji
verjetnostni gostoti za merjenje
lege. Vrtilna količina in energija sta
kvantizirani in zavzemata le
nezvezdne vrednosti, kot jih kažejo slike.